ChAdOx1 nCoV-19 Phase 1/2 Trial Results: Paracetamol Effects on Pain and Tenderness
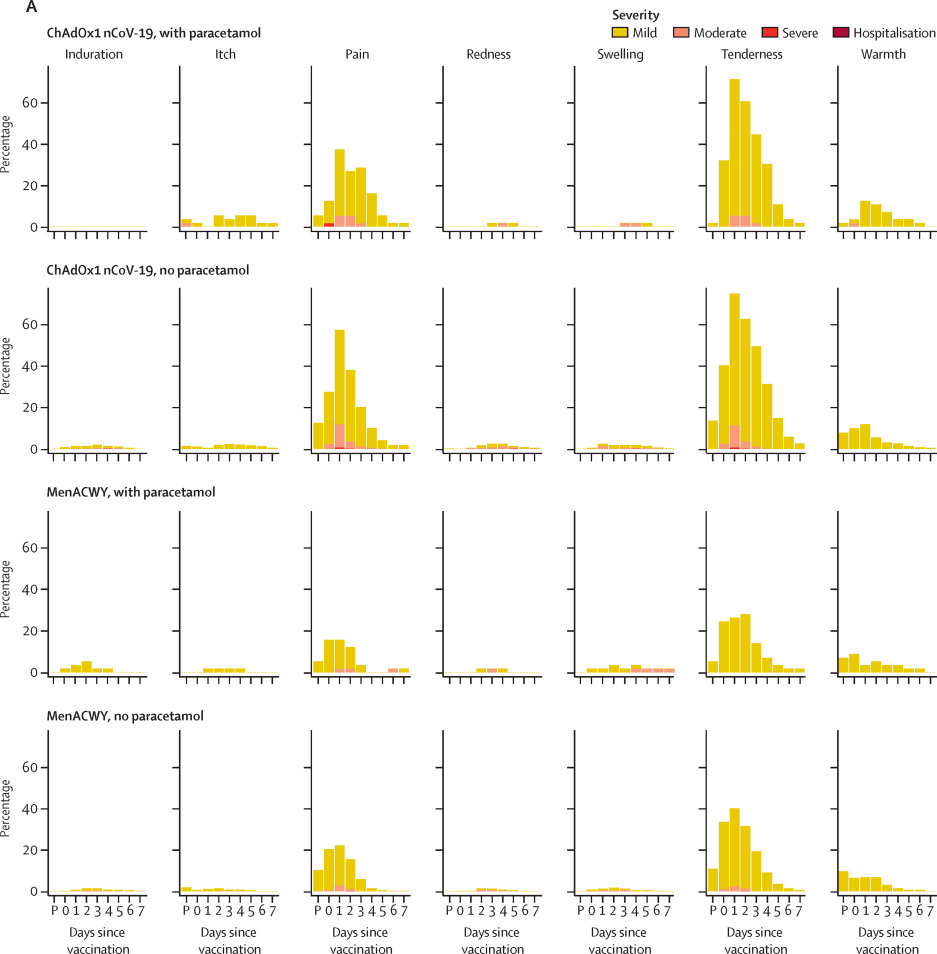
In a Phase 1/2 trial conducted between April 23 and May 21, 2020, participants were assigned to receive either the ChAdOx1 nCoV-19 or the MenACWY vaccine. A subset of participants (56 in the ChAdOx1 nCoV-19 group and 57 in the MenACWY group) took paracetamol. Participants who did not take paracetamol in both groups reported mild to moderate pain after vaccination. The use of paracetamol mitigated these local reactions: 50% of those receiving the ChAdOx1 nCoV-19 vaccine with paracetamol and 32% of those receiving the MenACWY vaccine with paracetamol reported less pain. Furthermore, less tenderness was reported in the groups taking paracetamol compared to those who did not.
0
0
Contributors are:
Who are from:
Tags
SARS-CoV-2 (COVID-19)
Biomedical engineering
Biomedical Sciences