Concept
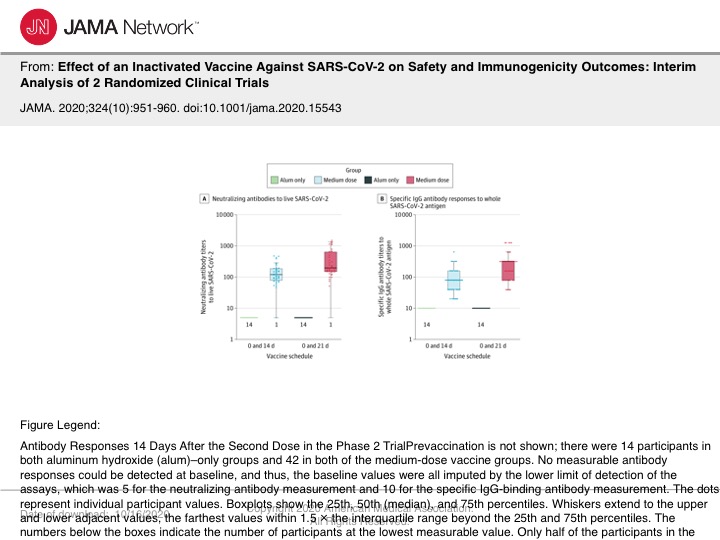
Figure 3. Antibody Responses 14 Days After the Second Dose in the Phase 2 Trial
0
1
Updated 2020-10-17
Tags
SARS-CoV-2 (COVID-19)
Biomedical Sciences
Related
Safety Outcomes for the Inactivated Whole Virus Vaccine by Wuhan Institute of Biological Products Phase II Trial
Immunogenicity Outcomes for the Inactivated Whole Virus Vaccine by Wuhan Institute of Biological Products Phase II Trial
Figure 3. Antibody Responses 14 Days After the Second Dose in the Phase 2 Trial
Wuhan Institute of Biological Products Vaccine Specifics (phases 1+2)