French open-label non-randomized clinical trial in support of treating COVID-19 patients with HCQ
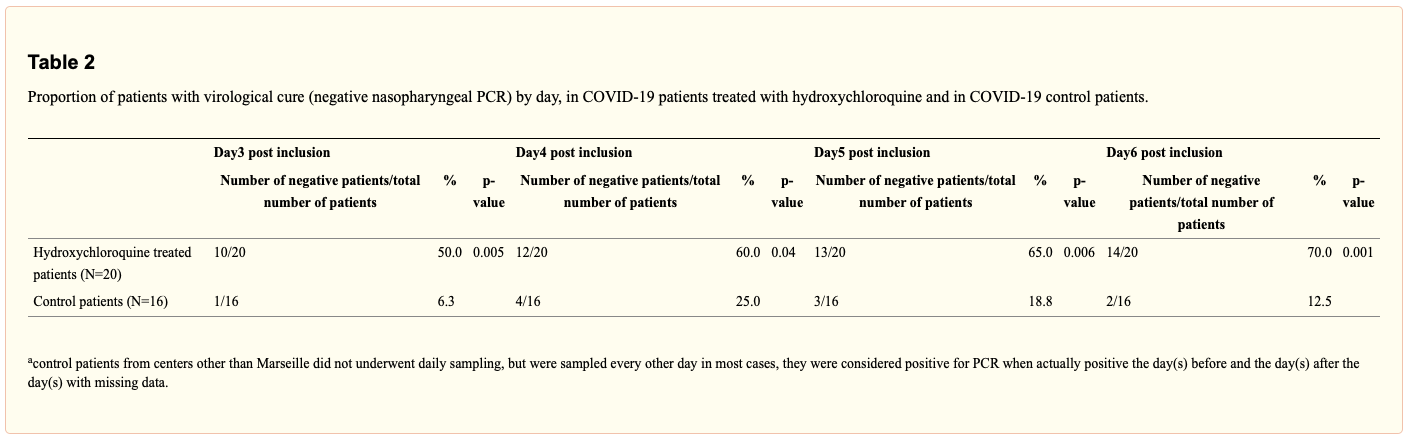
This study is the original report of using HCQ to treat COVID-19. In a sample of 26 patients, those treated with HCQ were administered 200 mg three times a day for 10 days. By day 3 of study, 10/20 of the patients given HCQ tested negative for COVID-19 compared to 1/16 not given HCQ. By day 5, there was 65.0% viral clearance in HCQ-treated patients as compared to 18.8% clearance in untreated patients. By day 6, 14/20 of those given HCQ tested negative compared to 2/16 not given HCQ. 6 patients were excluded from analysis due to symptom worsening or failure to follow-up, which makes the findings of this study difficult to assess.
0
1
Contributors are:
Who are from:
Tags
SARS-CoV-2 (COVID-19)
Biomedical Sciences
Related
French open-label non-randomized clinical trial in support of treating COVID-19 patients with HCQ
Efficacy of hydroxychloroquine in patients with COVID-19: results of a randomized clinical trial
Clinical and microbiological effect of a combination of hydroxychloroquine and azithromycin in 80 COVID-19 patients with at least a six-day follow up: A pilot observational study
In vitro testing revealed synergistic effect of combined hydroxychloroquine (HCQ) and azithromycin (AZM) on SARS-CoV-2