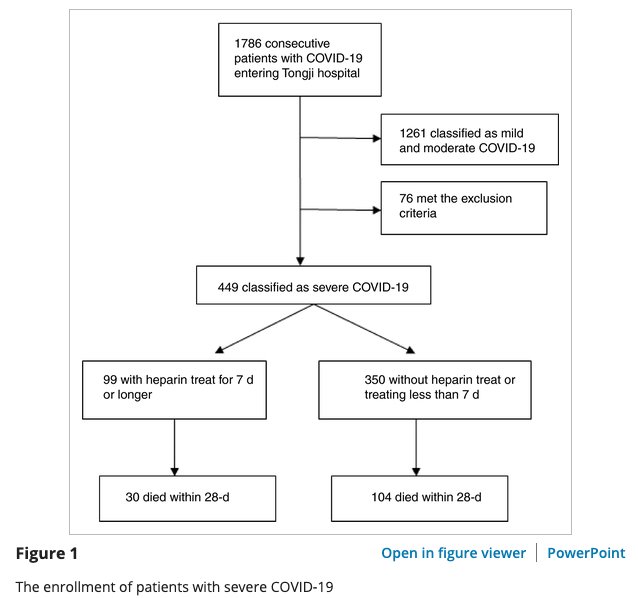
Inclusion criteria for evaluating heparin effectiveness in COVID-19 patients for patients at high risk of coagulopathy from retrospective study in Wuhan, China
-
admitted to Tongji Hospital of Huazhong University of Science and Technology in Wuhan from January 1 to February 13, 2020
-
tested positive for COVID-19 via RNA detection
-
"Severe COVID‐19 was defined as meeting any one of following items, according to the Diagnosis and Treatment Plan of COVID‐19 suggested by National Health Commission of China9: Respiratory rate ≥30 breaths/min; arterial oxygen saturation ≤93% at rest; PaO2/FiO2 ≤ 300 mm Hg."
-
treatment group had received unfractionated heparin or low molecular weight heparin (LMWH) for at least 7 days
0
1
Tags
SARS-CoV-2 (COVID-19)
Biomedical Sciences
Related
Inclusion criteria for evaluating heparin effectiveness in COVID-19 patients for patients at high risk of coagulopathy from retrospective study in Wuhan, China
Statistical analysis for evaluating heparin effectiveness in COVID-19 patients for patients at high risk of coagulopathy from retrospective study in Wuhan, China
SIC score and diagnostics for evaluating heparin effectiveness in COVID-19 patients for patients at high risk of coagulopathy from retrospective study in Wuhan, China